Cancer drug target identified
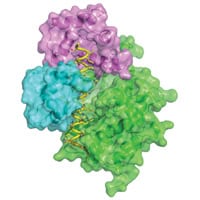
The molecular structure of a kinase involved in the development of cancer has been identified by scientists at the European Molecular Biology Laboratory using high-energy x-rays, and could have important applications in drug development.
The molecular structure of a kinase involved in the development of cancer has been identified by scientists at the European Molecular Biology Laboratory using high-energy x-rays, and could have important applications in drug development.

Calmodulin picks up the flood of calcium ions coming into the cell triggered by the cell’s signalling system. It binds to a particular part of DAPK, switching the kinase on so it can go and change the function of its targets. The team have determined which of DAPK’s amino acids are crucial for calmodulin to bind to.
“Faulty versions of DAPK are involved in the development of some cancers,” said Wilmanns, “So we want to know more about how this protein functions to allow its better exploitation as an anti-cancer target.”
DAPK has physical similarities to many other kinases controlled by calmodulin, meaning they are likely to interact in the same or similar ways. The three dimensional structure of the protein is useful to observe how kinases and calmodulin clip together and alter each other’s behaviour, and could provide a way for researchers to manipulate this interaction with drugs.
“That will provide a platform to get into drug discovery,” said Wilmanns, “Obviously, this is the beginning of the story.” Drugs to tackle neurodegeneration, cancer and cardiovascular disease may be developed.

