How much protein is in your beans?

Healthy high protein, low fat foods such as beans are becomingly increasingly prevalent in the food market with people taking a growing interest in their dietary health. Here we learn of a new protein level detection technique that has bean gaining acclaim
Healthy high protein, low fat foods such as beans are becomingly increasingly prevalent in the food market with people taking a growing interest in their dietary health. Here we learn of a new protein level detection technique that has bean gaining acclaim

The two key methods in identifying the amount of protein contained within foodstuffs are the Kjeldahl and Dumas combustion. The more traditional Kjeldahl method of protein/nitrogen determination has many recognised flaws whereas the lesser-known Dumas combustion method has been greatly improved with new pioneering technology making it a superior protein content analysis method. This article discusses the Kjeldahl method and investigates the Dumas combustion method as an improved alternative for measuring protein/nitrogen levels in beans.
Johan Kjeldahl, a Danish industrial chemist developed his nitrogen/ protein
 |
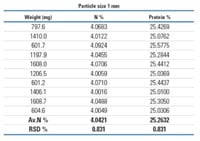
| Table 1: N/protein determination in beans |
The original Kjeldahl method involved large amounts of strong acids with very hot
 |
| Table 2: N/protein determination in red beans |
The protein/nitrogen determination method developed by Professor Jean-Baptiste
 |
| Table 3: N/protein determination in lentils |
Originating in academia this method was initially spurned by users in the industrial environment because of complications in achieving complete oxidation and contamination of the gaseous products with air. The comparative rapidity (minutes rather than hours), safety and economic advantages of the Dumas combustion method, as well as vast technological developments, have brought it back to the forefront of nitrogen/protein determination. It overcomes all concerns which tarnish the Kjeldahl method, including reduced sample preparation. This, in turn eliminates safety concerns, operator experience requirements, costs of materials and most significantly, the time involved.
Many official regulative organisations including the Association of Official Analytical Chemists (AOAC), the American Oil Chemist Society (AOCS) and the American Association of Cereal Chemists (AACC) support the new modified combustion method, as an alternative to the well-known Kjeldahl method. An example of this alternative method includes the Thermo Scientific FLASH 4000N/Protein analyzer, based on dynamic flash combustion, which features the safety and efficiency of the Dumas combustion method.
The experiment outlined below was carried out to validate new technologies, such as the Thermo Scientific FLASH 4000 analyzer and demonstrates the comparative efficacy to the Kjeldahl method.
The conditions of the experiment are detailed below with Figure 1 depicting the setup of instrumentation for the flash combustion method. The AOAC indicates that the suitable fineness of grind must be determined (for each different material analyzed) to achieve precision that gives RSD (equal or less) of 2% for ten successive determinations of nitrogen.
Temperature left reactor: 950 °C
Temperature right reactor: 840 °C
Temperature oven: 50 °C
Standard: 500 mg EDTA (9.59 %N)
Sample weight: 600 mg - 1.6 g
Different beans were chosen to validate the system in terms of accuracy and reproducibility according to the pre-treatment of the sample. The protein content is calculated automatically by the Thermo Scientific Eager Xperience Software using the default protein factor of 6.25. The protein factor can be changed in accordance with the food type.
Table 1 shows the analysis of ten consecutive determinations of bean samples with different homogenisation, 1mm and 2mm particle size. The data obtained indicate that it is not necessary for a fine homogenisation to reach a RDS% (equal or less) to 2% as suggested in the official methods.
Table 2 shows the nitrogen and protein data obtained from the red beans
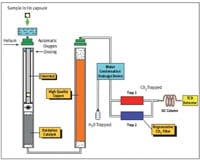 |
| Figure 1: Analytical Configuration within the Thermo Scientific FLASH 4000N/ Protein Analyser |
the above experiment demonstrates that current leading dynamic flash combustion instruments provide nitrogen/protein determination within a matter of minutes as opposed to hours. In addition the improved combustion method achieves this in a more environmentally-friendly manner than the Kjeldahl method with fewer safety concerns. Further benefits of these newer technologies include continuous operation through the use of automation and dual regenerating carbon dioxide filters operating alternately. The increased ease-of-use ensures that they do not require a specialist in the field to operate them.




