Therapeutic vaccine – is this the way forward for Parkinson’s treatment
Now that a potential molecular target has been elucidated, is it realistic to think in terms of a therapeutic vaccine for Parkinson’s disease? Certainly say Markus Mandler and Achim Schneeberger, but there is a problem – how to avoid autoimmunity? Parkinson’s disease (PD) is the second most common neurodegenerative disorder of the elderly (behind Alzheimer’s disease (AD)1; in people aged >65, its prevalence ranges between 100 and 250 cases per 100,000 in Western countries and 1,700 per 100,000 in China. It was long considered a motor disease characterised by akinesia/bradykinesia, rigidity and rest tremor. Its non-motor symptoms (neuropsychiatric, gastrointestinal, autonomous symptoms) are now well established2,3. PD treatments available to date primarily address the disease’s motor component. As they deliver symptomatic benefit only, they ultimately lose their clinical activity and fail3. Moreover, long-term use is associated with complications such as treatment induced dyskinesia. There are several areas of medical need. First, we lack a disease-modifying agent. Second, measures to combat treatment-induced dyskinesia are limited. Third, there are only a few therapeutic options for non-motor symptoms. Obviously, rational development of a disease-modifying therapeutic approach primarily requires understanding the underlying pathology. In the case of PD, there is mounting evidence for a causal and essential pathophysiological role of alpha-synuclein (aSyn). This field was pioneered by Spillantini and colleagues when they identified aSyn, by means of immunohistochemistry, to be a component of Lewy bodies4. Further arguments (see also Table 1) supporting a key role of aSyn in the pathogenesis of PD include the demonstration that certain dominantly inherited PD forms are caused by mutations in/duplications of the aSyn gene5-9, the observation that aSyn represents the major component of the neuropathological signature lesions in patients (Lewy bodies and Lewy neurites)10-12, the GWAS finding that certain aSyn gene variants contain the highest risk for sporadic PD13-16, the demonstration that overexpression of human aSyn recapitulates certain features of the disease in experimental animals17-21, and, finally, the fact that clinical symptoms and their progression correlate with the localisation and development of aSyn pathology11, 22. The latter is further supported by studies demonstrating pathological aSyn to be transmitted from cell to cell in a prion-like propagation pathway24. While all these studies suggest aSyn to be the toxic culprit, likely in the form of oligomers, downstream events such as mitochondrial dysfunction and oxidative stress are expected to mediate and even modulate its toxicity.

Table 1: aSynuclein is central to PD pathogenesis
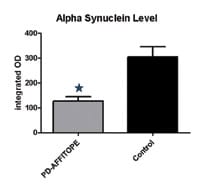
Groups of transgenic mice (PDGF-aSyn) received 6 s.c. immunizations using a PD-AFFITOPE-vaccine or vehicle. Brain homogenates were analyzed for aSyn burden by Western blot and levels were quantified densitometrically. Asterisk denotes p<0.05[/caption] Both of the above concerns can be dealt with by the AFFITOME technology, which uses short peptides mimicking the target sequence27. Key in the case of aSyn is to prevent the elicitation of antibodies reacting with other members of the synuclein family. This consists of three members – alpha, beta and gamma-synuclein – which do not only share extensive sequence homology but also a similar expression pattern. They are thought to act as chaperones thereby regulating vesicle transport and synaptic plasticity. Of note, aSyn knock-out mice are viable and show no overt neuronal phenotype indicating that aSyn function can be replaced by other synucleins. bSyn shows the highest overlap of expression with aSyn. It appears to counteract most of the toxic effects of aSyn. Its neuroprotective properties are in part mediated by AKT signalling. In addition, bSyn has been shown to prevent aggregation and oxidation of aSyn. Obviously, immunotherapeutic approaches need to respect these relations, in particular a vaccine targeting aSyn must not attack bSyn. In line with the above, the AFFiRiS PD drug development program focuses on AFFITOPEs eliciting Abs recognising aSyn while sparing bSyn. It delivered a series of candidates. These have been extensively tested in various transgenic preclinical models of synucleopathies28. AFFITOPE vaccination resulted in high target-specific (aSyn) antibody titres in plasma, antibodies were able to cross the blood brain barrier as evidenced by different techniques (in situ staining and CSF analysis). Antibodies elicited were shown to primarily recognise aSyn aggregates. Induction of these antibodies was associated with a lowering of cerebral aSyn burden, especially of its oligomeric form (see Fig 1). This was accompanied by a reduction of the degeneration of tyrosine hydroxylase expressing nerve cells. Most importantly, successfully immunised mice were also shown to exhibit improvements in motor as well as memory deficits (depending on the model system used). Studies to the mode of action indicate that clearance of aSyn involves the activation of microglia in the context of a milieu of anti-inflammatory cytokines28. Regarding safety, as expected, these peptide-protein conjugate vaccines do only elicit carrier-specific but not target-specific T cells. Thus, they avoid cellular autoimmunity. At the time points investigated, we do not have evidence for overt microgliosis, astrocytosis or a potential pathological inflammatory process. Together, these results demonstrate aSyn AFFITOPE vaccines to be safe on preclinical grounds and to exhibit disease-modifying properties.
- Schapira AH and Jenner P. Etiology and pathogenesis of Parkinson’s disease. Mov Diord. 2011; 26 (6): 1049-55.
- Poewe W. Non-motor symptoms in Parkinson’s disease. Eur J Neurol 2008; 15 (Suppl 1): 14-20.
- Stern MB, Lang A, Poewe W. Toward a redefinition of Parkinson’s disease. Mov Disorder 2012, 27 (1): 54-60.
- Spillantini MG; Schmidt ML; Lee VM et.al. Alpha-synuclein in Lewy bodies. Nature 1997;388:839-840
- Lansbury PT Jr; Brice A. Genetics of Parkinson's disease and biochemical studies of implicated gene products. Curr. Opin.Genet. Dev. 2002;12(3):299-6.
- Singleton AB; Farrer M; Johnson J et.al. alpha-Synuclein locus triplication causes Parkinson's disease. Science 2003;302:841.
- Polymeropoulos MH; Lavedan C; Leroy E et.al. Mutation in the alpha-synuclein gene identified in families with Parkinson's disease. Science 1997;276:2045-2047.
- Ross OA; Braithwaite AT; Skipper LM et.al. Genomic investigation of alpha-synuclein multiplication and parkinsonism. Ann.Neurol. 2008;63(6):743-50.
- Eriksen JL; Dawson TM; Dickson DW et.al. Caught in the act: alpha-synuclein is the culprit in Parkinson's disease. Neuron 2003;40:453-56.
- Braak H; Del Tredici K; Rüb U et.al. Staging of brain pathology related to sporadic Parkinson's disease. Neurobiol Aging,2003;24:197-11.
- Jellinger KA. Synuclein deposition and non-motor symptoms in Parkinson disease. J. Neurol. Sci. 2011;15;310(1-2):107-11.
- Dickson DW; Fujishiro H; Orr C et.al. Neuropathology of non-motor features of Parkinsons disease. Parkinsonism Rel Disord,2009;15(S3):S1-S5.
- Gandhi S; Wood NW. Genome-wide association studies: the key to unlocking neurodegeneration? Nat. Neurosci.2010;13(7):789-94.
- Simón-Sánchez, J; Schulte C; Bras JM et.al. Genome-wide association study reveals genetic risk underlying Parkinson's disease. Nat. Genet. 2009;41(12):1308-1312.
- Satake, W; Nakabayashi Y; Mizuta I et.al. Genome-wide association study identifies common variants at four loci as genetic risk factors for Parkinson's disease. Nat. Genet. 2009;41(12):1303-1307.
- Edwards TL; Scott WK, Almonte C et.al. Genome-wide association study confirms SNPs in SNCA and the MAPT region as common risk factors for Parkinson disease. Ann Hum Genet 2010;74(2):97-109.
- Masliah E; Rockenstein E; Veinbergs I et al. Dopaminergic loss and inclusion body formation in alpha-synuclein mice:implications for neurodegenerative disorders. Science 2000;287:1265-1269.
- Lotharius J; Brundin P. Pathogenesis of Parkinson's disease: dopamine, vesicles and alpha-synuclein. Nat Rev Neurosci.2002;3(12):932-942.
- Fleming SM; Salcedo J; Fernagut PO et al. Early and progressive sensorimotor anomalies in mice overexpressing wild-type human alpha-synuclein. J Neurosci. 2004;24(42):9434-9440.
- Fleming SM; Tetreault NA; Mulligan CK et al. Olfactory deficits in mice overexpressing human wildtype alpha-synuclein. Eur J Neurosci. 2008;28(2):247-256.
- Rockenstein E; Crews L; Masliah E. Transgenic animal models of neurodegenerative diseases and their application to treatment development. Adv Drug Deliv Rev 2007; 59(11):1093-102.
- Lim S-Y; Fox S.H.; Lang A.E. Overview of the extranigral aspects of Parkinson Disease. Arch Neurol. 2009;66(2):167-172.
- Savitt JM; Dawson VL; Dawson TM. Diagnosis and treatment of Parkinson disease: molecules to medicine. J Clin Invest.2006; 116(7):1744-54.
- Luk KC et al. Pathological alpha-synclein transmission initiates Parkinson-like neurodegeneration in nontransgenic mice. Science 2012; 338:949-53.
- Spillantini MG, Goedert M. The alpha-synucleopathies: Parkinson’s disease, dementia with Lewy bodies and multiple system atrophy. Ann NY Acad Sci 2000; 920: 16-27.
- Masliah Masliah E, Rockenstein E, Adame A, Alford M, Crews L, Hashimoto M, Seubert P, Lee M, Goldstein J, Chilcote T, Games D, Schenk D (2005) Effects of alpha-synuclein immunization in a mouse model of Parkinson’s disease. Neuron 46(6):857–868.
- Schneeberger A, Mandler M, Mattner F, Schmidt W (2010) AFFITOME technology in neurodegenerative diseases: the doubling advantage. Hum Vacc 6(11):948–952.
- Mandler M, Valera E, Rockenstein E, Weninger H, Patrick C, Adame A, Santic R, Meindl S, Vigl B, Smrzka O, Schneeberger A, Mattner F, Masliah E. Next-generation active immunization approach for synucleinopathies: implications for Parkinson's disease clinical trials. Acta Neuropathol. 2014 Jun;127(6):861-79.
- Schneeberger A, Mandler M, Mattner F, Schmidt W (2012) Vaccination for Parkinson’s disease. Parkinsonism Relat Disord 18(Suppl 1):S11–S13.




